Environment & Energy
Related: About this forumA Uranium Adsorbing Alginate Polyacylonitrile Membrane With Superior Regeneration Performance.
The paper I'll briefly discuss is this one:
Preparation of Amidoxime Polyacrylonitrile and Sodium Alginate Composite Membrane with Superior Regeneration Performance for Efficient Uranium Adsorption Yunyang Gui, Guoyan Qi, Nannan Xie, Qiufang Li, Xiaoli Su, Yang-Hai Zheng, Hui Ruan, Yongde Yan, Yun Xue, Sheng Wu, and Fuqiu Ma Industrial & Engineering Chemistry Research 2026 65 (13), 7108-7119
In the presence of an oxygen atmosphere, uranium supplies on the planet Earth are inexhaustible. There is no technology that can consume all of this mildly radioactive element - the decay series of which supplies the bulk of the internal heat of the planet - although members of its decay series, the formation of which can be prevented by fissioning the parent nuclide, are highly radioactive. Uranium is continuously cycled through the Earth's crust from the mantle by volcanism and continental uplift, where erosion causes it to be, given its low but significant solubility in the +6 valence state, generally is a series of acquo complexes related to the UO22+ ion, a U(VI) species, washed to the sea. The uranium that is washed by rivers into the ocean, accumulates until the solubility is exceeded, whereupon it precipitates and is returned to the mantle by subduction.
The uranium content of the world's major rivers has been measured and is recorded in this publication and many other publications:
François Chabaux, Jean Riotte, Norbert Clauer, Christian France-Lanord, Isotopic tracing of the dissolved U fluxes of Himalayan rivers: implications for present and past U budgets of the Ganges-Brahmaputra system, Geochimica et Cosmochimica Acta, Volume 65, Issue 19, 2001, Pages 3201-3217.
A table from that paper:
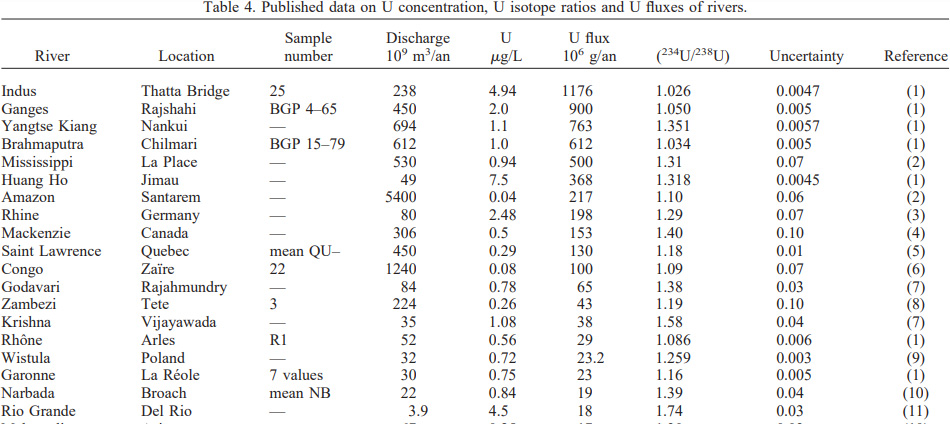
The output of the Ganges river system, 1176 metric tons (1176 X 106 grams) The amount of uranium released into the Indus river, converted to plutonium by neutron capture in nuclear reactors, is equal to close to 100 Exajoules of primary energy each year.
Overall, it is now understood that uranium content of the Earth's oceans is roughly 4.5 billion tons, at a concentration of about 3.3-3.4 micrograms per liter. Captured and converted to plutonium, this is the equivalent of over 500,000 years of human energy consumption at a prodigious energy demand of 700 Exjaoules/year, 50 Exajoules more than we are consuming as of now from all sources of energy, including the largest source of energy, about which antinukes and "I'm not an antinuke" antinukes couldn't care less, dangerous and deadly fossil fuels, which are destroying the planetary atmosphere.
It is true that almost all of the Earth's major riverine and riparian systems have been sacrificed to so called "renewable energy" - a scheme to industrialize all wilderness systems from rivers, deserts, mountain tops and even the seas themselves, so people can continue to worship their cars, go to "no nukes" rock concerts, and log on the internet to see "influencers" - whatever the hell they are - and thus the recharge from rivers is limited in these times. I have argued however, in a sample case of California, that it might be possible to restore riverine systems to free flow along with other bodies of water via supercritical water desalination:
The Energy Required to Supply California's Water with Zero Discharge Supercritical Desalination.
In addition, we are mining fossil groundwater all over the world, and as ground water flows to granite and other minerals with significant uranium content, many of these water supplies are contaminated with NORM (Naturally Occurring Radioactive Materials) most prominently with uranium, but also its radioactive decay series products. The flowback water from fracking operations in Pennsylvania is far more radioactive than the trivial tritium concentrations released at Fukushima about which all of our dumb antinukes carry on, but again, antinukes and "I'm not an antinuke" antinukes couldn't care less about fossil fuels, even when they release vast amounts of radioactivity. (Coal ash is a significant source of uranium.)
Although uranium is ubitiquous, and its native radioactivity - excluding, again, decay products - is low owing to its long half-life, thus meaning it isn't very dangerous from a radiological sense, it is a chemotoxin, leading to kidney damage. Thus removing uranium from mined groundwater is a good idea, making it a source of uranium for energy purposes.
This brings me to the paper cited at the outset, which begins with some statements I personally think are nonsense, irrespective of the fine paper's and introduction's other content.
The introductory text:
Substantial advancements have been made in the field of uranium adsorption by using a variety of functional materials. Notable progress has been reported for systems based on inorganic compounds, (11) synthetic polymers, (12) naturally derived biomass, (13) metal–organic frameworks, (14) covalent organic frameworks, (15) and ion exchange resins. (16) Among them, membrane-based polymer materials have often been used as adsorbents due to their advantageous properties, including facile synthesis, modification, and chemical stability. In particular, the superior coordination affinity of amidoxime (AO) toward U(VI) made membrane materials containing such groups widely used for uranium adsorption. (17,18) For instance, Chen et al. (19) synthesized a polydopamine–amidoxime polyacrylonitrile membrane through a combination of dopamine polymerization/deposition and nonsolvent-induced phase separation. This membrane exhibited a remarkable uranium adsorption efficiency and selectivity. In another study, Sun et al. (20) fabricated an ultrathin PAO-based membrane by incorporating quaternized chitosan and PAO into a cross-linked sulfonated cellulose nanocrystal matrix, demonstrating excellent antibiofouling properties and significantly enhanced uranium adsorption capacity.
Concurrently, in response to growing environmental concerns, the biodegradability of polymer adsorbent materials is gradually gaining attention. (21) Sodium alginate (SA), as a natural polysaccharide, is particularly attractive due to its biodegradability, biocompatibility, and renewability and is rich in functional modifying groups such as carboxyl and hydroxyl groups. Its excellent film-forming ability and structural stability make it a favorable candidate for material synthesis...]
Alginate is a biologically sourced material, obtained from seaweed; acrylonitrile is a petroleum product. Theoretically, although not practically in the current world, acrylonitrile can be synthesized from biological materials and/or via a methanol based pathway via the hydrogenation of carbon dioxide or graphite produced via the Boudouard reaction driven by nuclear heat.
This is hardly the first paper written about pathways to extract uranium from seawater, groundwater, or process water using amidoximine functionalized polymers; there are literally thousands of such papers, going back over decades, but the paper claims the process is more selective than many other options, and is readily subject to reuse.
Some graphics from the paper:
A synthetic scheme cartoon from the abstract:
Another synthetic scheme cartoon from inside the paper:
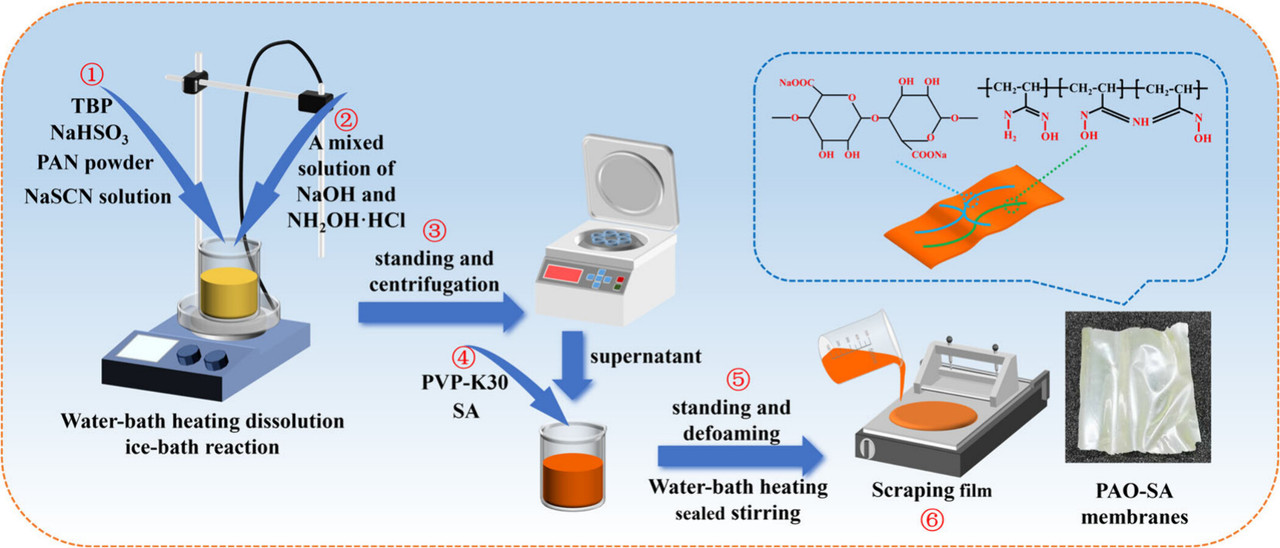
The caption:
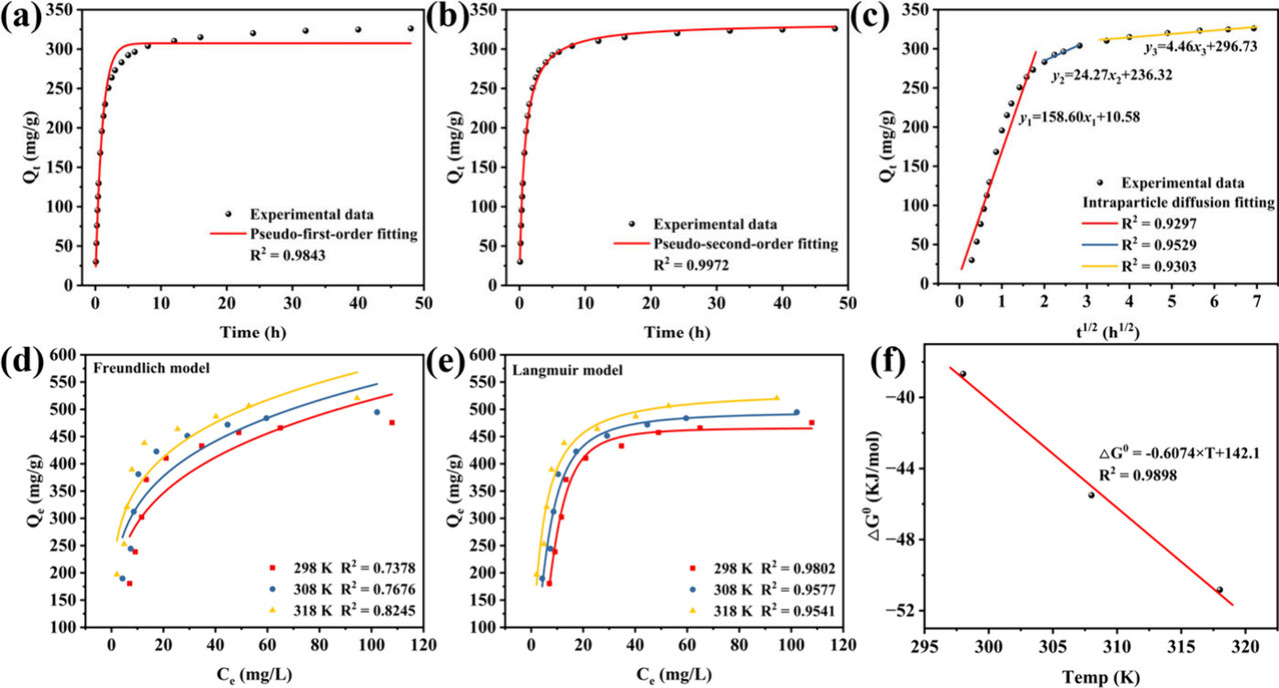
The caption:
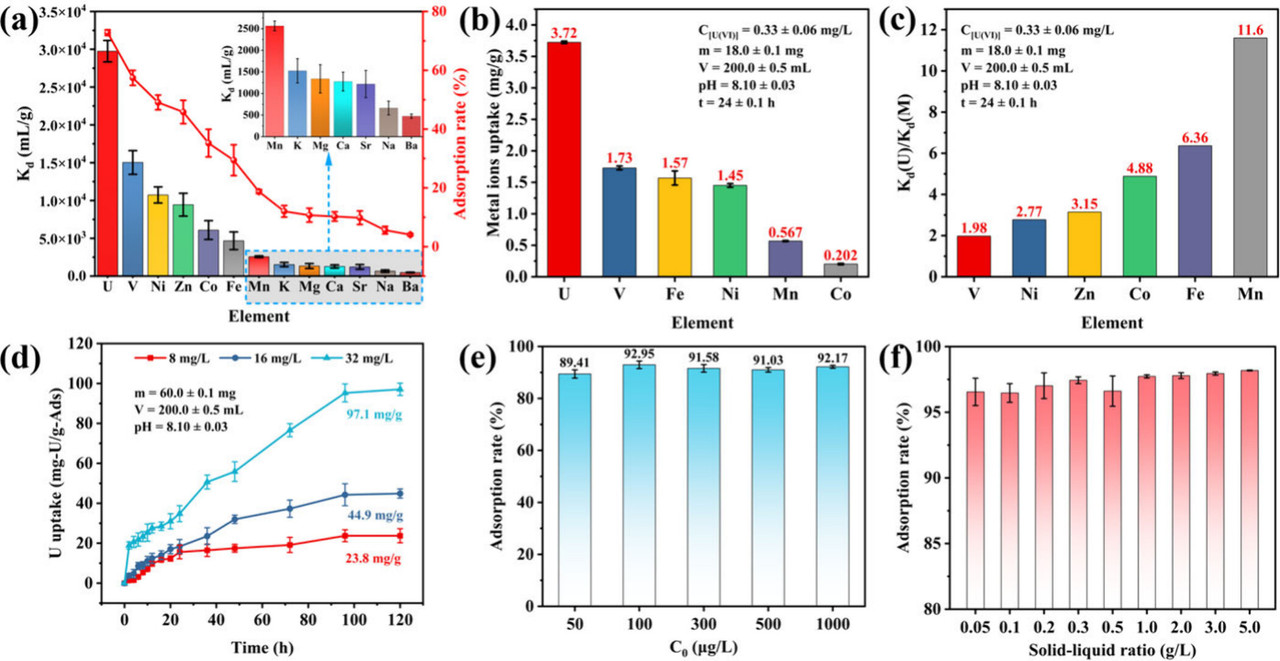
The caption:
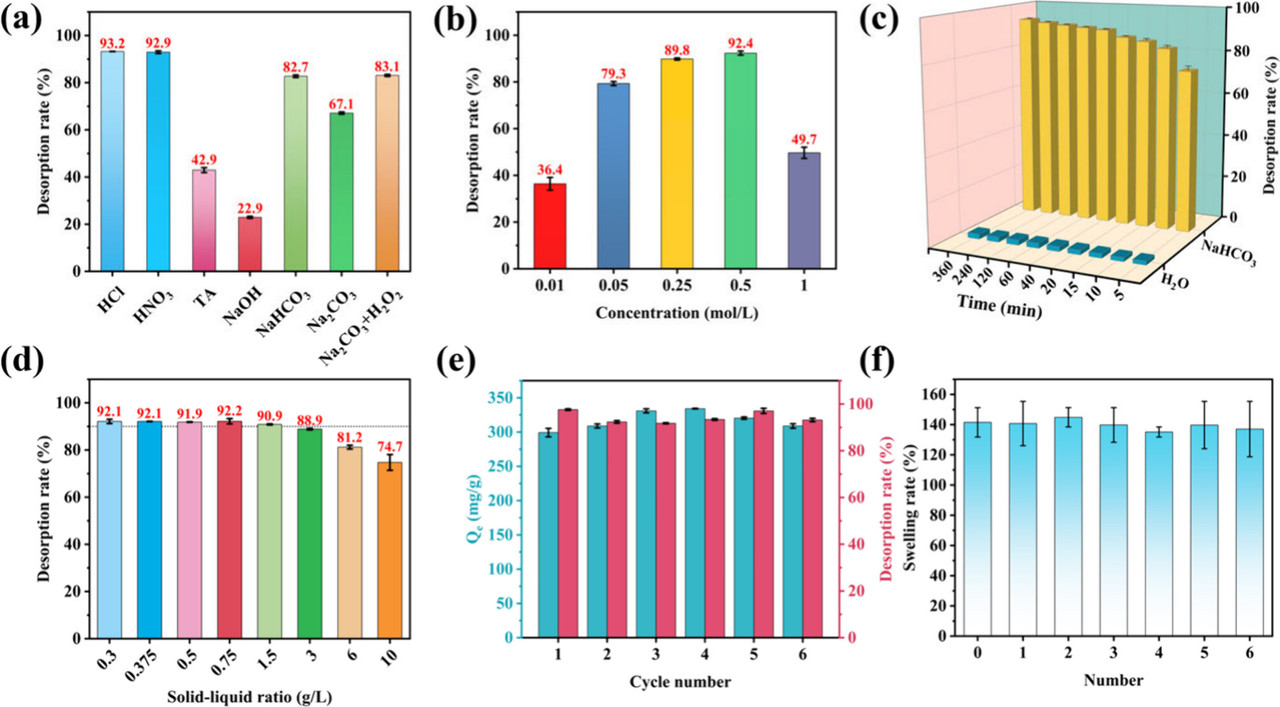
The caption:
A table from the text comparing the performance of the authors' product with a small selection of others from the literature:
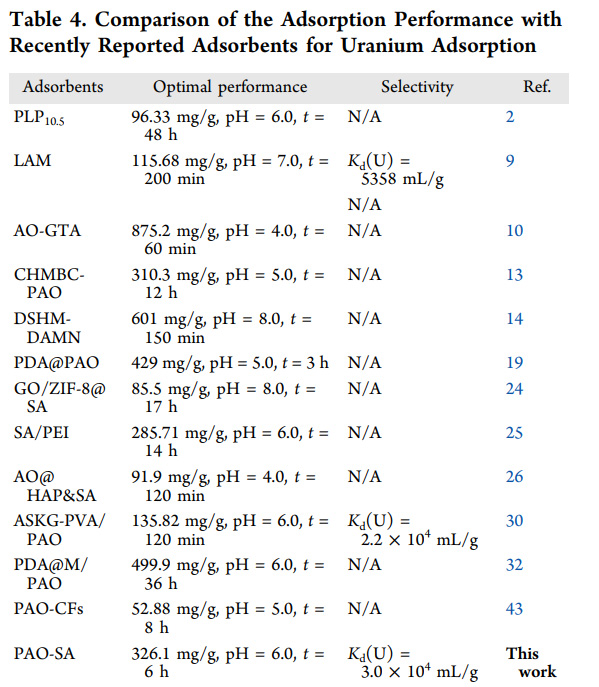
I have argued, and will continue to argue, that with incorporation of fast neutron spectrum nuclear reactors as well as adjustment of CANDU type thermal reactors incorporating ternary uranium, thorium, plutonium fuels, the uranium already mined, as well as the thorium dumped in tailings from lanthanide mines that serve, among other things, to support the useless wind industry, that humanity could provide all of its energy needs - eliminating all oil, gas, coal mines, as well as restoring wilderness industrialized for useless so called "renewable energy" junk - for centuries.
Nevertheless, products as those described in this paper (and many other papers), can help clean up naturally uranium and NORM contaminated groundwater, fracking wastewater as well as process water from uranium processing plants. (I personally do not like extraction procedures for nuclear fuel processing, and greatly prefer the fluoride volatility type and related pyroprocessing schemes.) The uranium so removed from these sources can then be available as fuels.
From the paper's conclusion:
(With reference to the U to V ratio, vanadium is always recovered as a side product of these procedures.)
Have a nice Mother's day, if you are celebrating it with members of your family.
Shipwack
(3,099 posts)The anti renewable (solar, wind, hydropower) remarks are from you, and not the paper? Or both?
NNadir
(38,476 posts)...so called "renewable energy," since, unlike the modern members of the Sierra Club, who never see a wilderness they don't wish to convert to an industrial park for solar and wind, my environmental views are consistent with those of the Sierra Club's founder John Muir, who founded the Club in a failed attempt to prevent industrialization of a natural wilderness, the Hetch Hetchy valley in Yosemite National Park, now a hydroelectric plant.
Nuclear energy is the only form of energy I regard as clean and sustainable
I know this view is unpopular in some circles here, but I unrepentantly hold this view based on roughly 40 years of consideration ever since the Chornobyl reactor exploded, establishing nuclear's worst case.
Thanks for asking.
Shipwack
(3,099 posts)Envirogal
(322 posts)Never can answer the nuclear waste issue though. Wouldn’t have a problem putting a nuclear plant at the Hetch Hetchy to cool these monstrosities though, right?
Hetch Hetchy is a gorgeous miraculous place that Muir saved. He also saved the Grand Canyon from development. But there are all kinds of lands and waters that were not saved from mining and other infrastructure projects. So your argument using the Trojan Horse of Muir before nuclear energy was invented, is a Stretch Stretchy.
Solar rooftops do not disrupt the wilderness. And using John Muir as an example is a bit much given how much of our natural world, including the rivers to build behemoth concrete nuclear plants, that Muir would most likely also oppose. Or the delicate desert ecosystems to store their waste.
Every energy tech has pros and cons—all have an impact, especially trying to satisfy the needs of a growing 8.3 BILLION population.
I’m still waiting for that Chernobyl- like fallout from rooftop solar panels to where the land the size of Rhode Island will not be habitable for thousands of years.